OpenAI Enters the AI-Bio Arms Race with GPT-Rosalind

OpenAI has officially unveiled GPT-Rosalind — its first frontier reasoning model purpose-built for biology, drug discovery, and translational medicine. Named after the British chemist Rosalind Franklin, whose X-ray diffraction work was foundational to the discovery of DNA’s double helix, the model arrives just two days after OpenAI’s strategic partnership with Novo Nordisk and one day after AWS’s launch of Amazon Bio Discovery — making the second week of April 2026 arguably the most consequential stretch yet for AI in pharma.
GPT-Rosalind is not being released into the wild yet with only a few researchers and institutions being invited to test it behind a Trusted Access program. Available initially only to qualified Enterprise customers in the United States via ChatGPT, Codex, and the API. Launch partners include Amgen, Moderna, the Allen Institute, and Thermo Fisher Scientific, with ongoing collaborations at Los Alamos National Laboratory on AI-guided protein and catalyst design. The model is the first in what OpenAI describes as a long-term “Life Sciences model series.”
This is OpenAI essentially explicitly conceding that general-purpose models — even GPT-5 class systems — are not enough for serious biological research and are prone to hallucinations and misinformation. Domain-specific reasoning, tool use over scientific databases/papers, and governance built for regulated environments are now table stakes.
What GPT-Rosalind is Meant to Do?
Rather than being marketed as a chatbot for scientists, GPT-Rosalind is positioned as a research workflow engine. According to OpenAI’s announcement, the model is optimized for the kind of multi-step, tool-heavy work that occupies most of a discovery scientist’s day:
- Target discovery and validation across genomics, proteomics, and disease biology
- Literature synthesis across millions of papers and structured databases
- Hypothesis generation with multi-step biological reasoning
- Sequence-to-function interpretation for proteins and nucleic acids
- Experimental planning and analysis of omics data
The model is trained to reason across molecules, proteins, genes, pathways, and disease-relevant biology — and OpenAI claims it is significantly better than its mainline models at orchestrating calls to scientific tools and databases through long-horizon workflows. Crucially, the company is repeating the line every life sciences AI vendor now has to repeat: it is not designed to replace expert judgment or real-world experimental validation, with humans essential for the research loop.
Performance Benchmarks: 95th Percentile on Sequence Prediction
As per the press briefing, the most striking performance signal came from a partnership with Dyno Therapeutics, an AAV gene therapy company. In an evaluation using unpublished, “uncontaminated” RNA sequences — meaning data the model could not have seen during training — GPT-Rosalind was given a sequence-to-function prediction and generation task. When evaluated with best-of-ten model submissions, the results were genuinely surprising:
- Above the 95th percentile of human experts on the prediction task
- Around the 84th percentile of human experts on the sequence generation task
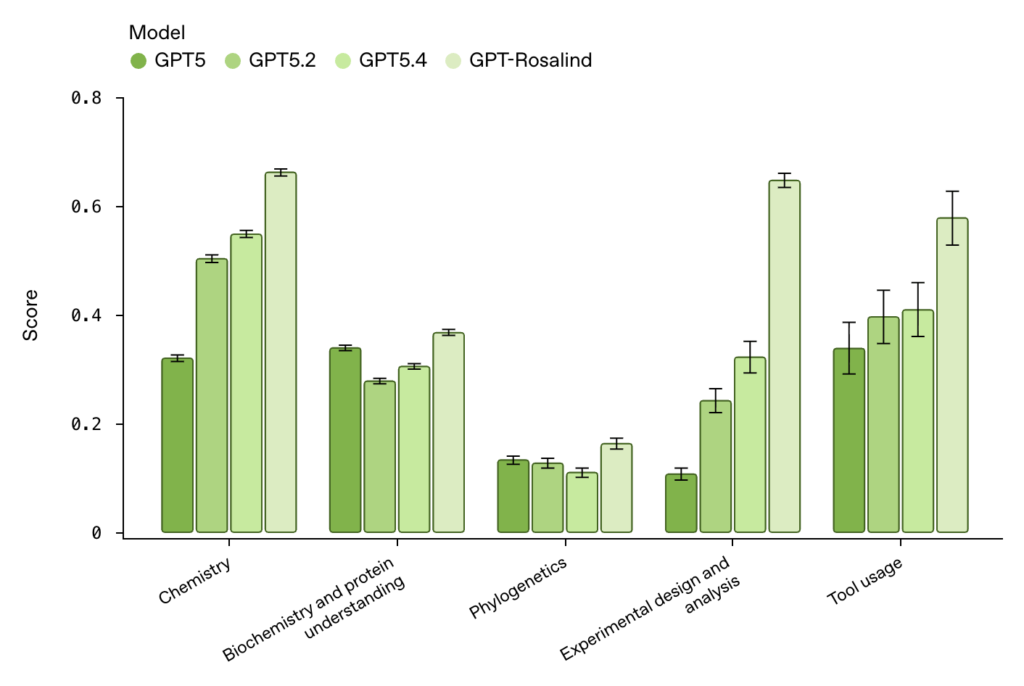
These numbers were measured against 57 historical scores from human experts in the AI-bio field, as reported in VentureBeat’s launch coverage. Whether they generalise outside the Dyno setup is the question every benchmark eventually faces — but it is a meaningful early signal that a domain-specialised reasoning model can match or exceed the median expert on at least some non-trivial bio tasks. Compared to its own models, GPT-Rosalind performed better across different fields such as chemistry, phylogenetics, tool usage, etc. The biggest difference was seen in experimental design and analysis, a huge factor in choosing GPT-Rosalind over the other models.
The Codex Plugin: 50+ Tools, Freely Available
Alongside the gated model, OpenAI is releasing something far more open: a Life Sciences research plugin for Codex, available immediately on GitHub. The plugin connects models to over 50 scientific tools and data sources spanning:
- Human genetics
- Functional genomics
- Protein structure
- Biochemistry
- Clinical evidence
- Public study discovery
Eligible Enterprise users get the plugin paired with GPT-Rosalind for deeper biological reasoning. Everyone else can use the same plugin package against OpenAI’s mainline models. This integration suggests OpenAI’s long-term moat in life sciences will live as much in the ecosystem of connectors as in the model weights themselves. It is the same playbook AWS used in launching Amazon Bio Discovery: own the integration layer, then sell access to whichever model wins.
Trusted Access: Biosafety as a Product Feature
GPT-Rosalind’s deployment structure is itself a statement. Rather than a broad rollout, OpenAI is requiring organisations to undergo a qualification and safety review demonstrating:
- Legitimate research use cases with public health benefit
- Robust governance and misconduct reporting
- Compliance with specific life sciences research preview terms
- Enterprise-grade access management
As Axios reported earlier this year, more than 100 reputed scientists have called for tighter controls on sensitive biological data used to train AI systems, citing dual-use concerns around models that can redesign biological structures. A model that can be used to design therapeutic proteins can, in principle, be misused to design dangerous ones. OpenAI’s “trusted-access deployment structure” is the company’s bet that controlled enterprise rollout — not open weights — is the only defensible path for capable bio-AI in 2026.
Competitive Landscape: GPT-Rosalind Walks Into a Crowded Room
The launch lands in what is now an unmistakably crowded field. The AI-for-biology stack has fragmented into clear archetypes — and GPT-Rosalind sits awkwardly across several of them.
1. Hyperscaler infrastructure plays — AWS, Microsoft, Google
Just one day before GPT-Rosalind, AWS launched Amazon Bio Discovery, an agentic platform that unifies biological data ingestion, AI model selection, and CRO-mediated wet-lab testing into a single cloud-native loop. Labcritics covered the launch in detail in Amazon Bio Discovery: Latest Entrant Aiming to Rewire the Antibody Discovery Pipeline. AWS’s bet is on infrastructure breadth and CRO integration. OpenAI’s bet is on reasoning depth.
Microsoft has Copilot Health and a separate angle through its OpenAI investment. Google’s bet runs through Isomorphic Labs.
2. AI-first drug discovery firms — Isomorphic Labs, Recursion, Insilico
Alphabet’s Isomorphic Labs has been quietly turning AlphaFold-class capability into pharma revenue. In April 2025, Isomorphic closed major collaboration deals with Eli Lilly and Novartis worth nearly $90 million upfront, and in February 2026 unveiled a new drug design engine intended to outdo AlphaFold 3. Recursion, Insilico Medicine, Schrödinger, and Exscientia all play in adjacent territory.
The key difference: Isomorphic and its peers are building drug pipelines. OpenAI is building a reasoning model that pipelines can plug into.
3. GPU-accelerated biology — NVIDIA BioNeMo
NVIDIA’s BioNeMo framework, launched in late 2024, offers pre-trained models like ESM-2 and Geneformer along with GPU-optimised infrastructure for protein folding, single-cell analysis, and molecular generation. NVIDIA owns the compute layer most life sciences AI runs on — including, almost certainly, the silicon GPT-Rosalind itself was trained on.
4. Autonomous and self-driving labs — Lila, Medra
A different bet entirely is being made by companies trying to remove the human from the loop altogether. Flagship Pioneering’s Lila Sciences is building an AI “scientist” capable of generating hypotheses, designing experiments, and interpreting results without human intervention. Medra.ai, fresh off a $52M round, is doing similar work with closed-loop robotic experimentation, as Labcritics reported in Medra.ai Gets a Boost to Foresee the Future of AI Drug Discovery.
GPT-Rosalind is, by contrast, deliberately and explicitly not trying to replace the scientist. It is a reasoning layer for human researchers, not an autonomous agent for empty labs.
5. Vertical SaaS — Benchling, Certara
Application-layer players like Benchling and Certara.AI continue to dominate scientist-facing workflow tooling. They are likely to integrate models like GPT-Rosalind rather than compete with them — particularly if OpenAI’s plugin architecture continues to mature.
Why OpenAI Is Doing This Now
Three forces converge to explain the timing.
First, customer pull. OpenAI already has deep relationships with pharma. Eli Lilly struck an OpenAI collaboration in 2024 on antibiotics for drug-resistant bacteria. Sanofi and Formation Bio are working with OpenAI on clinical trial enrollment. Moderna has a wide-ranging partnership. Thermo Fisher is using OpenAI to accelerate drug development. And as of April 14, 2026, Novo Nordisk has signed on for end-to-end deployment across R&D, manufacturing, and commercial operations, with pilot programs running through 2026. These customers were going to demand a domain-specialised model whether OpenAI built one or not.
Second, competitive pressure. AWS Bio Discovery, Isomorphic, NVIDIA, Anthropic’s Claude for Healthcare, and a long tail of AI-first biotechs are all moving on the same opportunity. Domain-specific models, as Futurum Group’s analysis noted, are increasingly seen as AI’s next competitive battleground after general-purpose chat.
Third, the economics of pharma R&D. The traditional drug discovery timeline runs 10–15 years from target to approval at an average cost of around $2 billion. Even modest gains at the earliest stages — better target selection, stronger hypotheses, fewer dead ends — compound into substantial savings downstream. McKinsey has previously estimated AI could unlock $60–110 billion per year for the pharma and medtech sectors. That number is now driving investor and partnership behaviour at every layer of the stack.
Open Questions
GPT-Rosalind launches with momentum, but the open questions are real:
- Reliability vs hallucination. AI agent reliability and hallucination management is the single largest cited adoption blocker in enterprise AI. In a domain where a wrong answer can mean a failed trial or a misallocated billion-dollar program, the bar is brutal.
- Generalisation beyond Dyno. The 95th-percentile result was on a partner-curated benchmark. Will the same performance hold across protein engineering, small-molecule design, and clinical-evidence reasoning?
- Defensibility of the moat. Anthropic, Google, and open-weights competitors will catch up on raw capability. The plugin ecosystem and customer entrenchment matter more than the model.
- Regulatory posture. No fully AI-discovered drug has cleared Phase 3. Regulatory acceptance of AI-generated hypotheses, experimental designs, and submissions remains nascent.
- Trusted Access at scale. Restricting access to qualified U.S. Enterprise customers protects against misuse but caps adoption. Loosening that constraint without weakening biosafety is the harder long-term problem.
Outlook
GPT-Rosalind is best read as the first domain-specialised frontier model from a major lab — not the last. The age of one-model-fits-all generative AI is closing. What replaces it is a stack of vertical reasoning models, each with a framework that is tuned for a domain’s tools, databases, evaluation criteria, and risk profile. Life sciences was always going to be the first vertical to get its own purpose-built frontier model, because the stakes are highest, the data is richest, and the customers are most willing to pay.
The strategic question for the rest of 2026 is no longer whether AI will reshape drug discovery — that is settled. The questions are which layer of the stack creates the most defensible economics, whether the biggest gains accrue to the model labs (OpenAI, Anthropic, Google), the hyperscalers (AWS, Azure, GCP), the vertical AI biotechs (Isomorphic, Recursion, Insilico, Absci), or the autonomous lab companies (Lila, Medra) — and whether, after a decade of hype, an AI-discovered drug finally clears Phase 3 drug trials. GPT-Rosalind does not answer those questions, however it makes the answer matter more, sooner.



